Endotronix, Inc., a digital health and medical technology company dedicated to advancing the treatment of heart failure (HF), today announced its PROACTIVE-HF pivotal study successfully completed enrollment. Designed to evaluate the safety and efficacy of the Cordella™ Pulmonary Artery (PA) Sensor, the data will support the pre-market approval (PMA) application for market access in the U.S.
“Building upon a strong foundation of evidence in favor of PA pressure-guided therapy, the PROACTIVE-HF trial is investigating a more comprehensive clinical picture for both clinicians and patients,” commented Liviu Klein, MD, Section Chief of Advanced Heart Failure, Mechanical Circulatory Support, Pulmonary Hypertension, and Heart Transplant at the University of California San Francisco and national principal investigator of the PROACTIVE-HF trial. “Cordella provides daily PA pressure and vital sign data to help clarify patient’s clinical status. Additionally, we are seeing high levels of engagement and lifestyle changes as a result of patients having visibility to their own data. We look forward to sharing the study results in the coming months.”
Notably the first global investigational device exemption (IDE) study for PA pressure-guided HF management, PROACTIVE-HF enrolled over 450 patients across the U.S., Ireland, and Belgium. Primary endpoints for the single-arm, multi-center trial include mortality and HF hospitalizations as well as device safety. The company expects to report primary endpoint data from the trial in the first half of 2024.
“I would like to congratulate all our clinical partners on their tremendous effort to complete enrollment for PROACTIVE-HF. Their hard work and dedication are key to improving outcomes and expanding patient access, and we appreciate their continued commitment to this trial,” stated Harry Rowland, CEO and co-founder of Endotronix.
About Endotronix
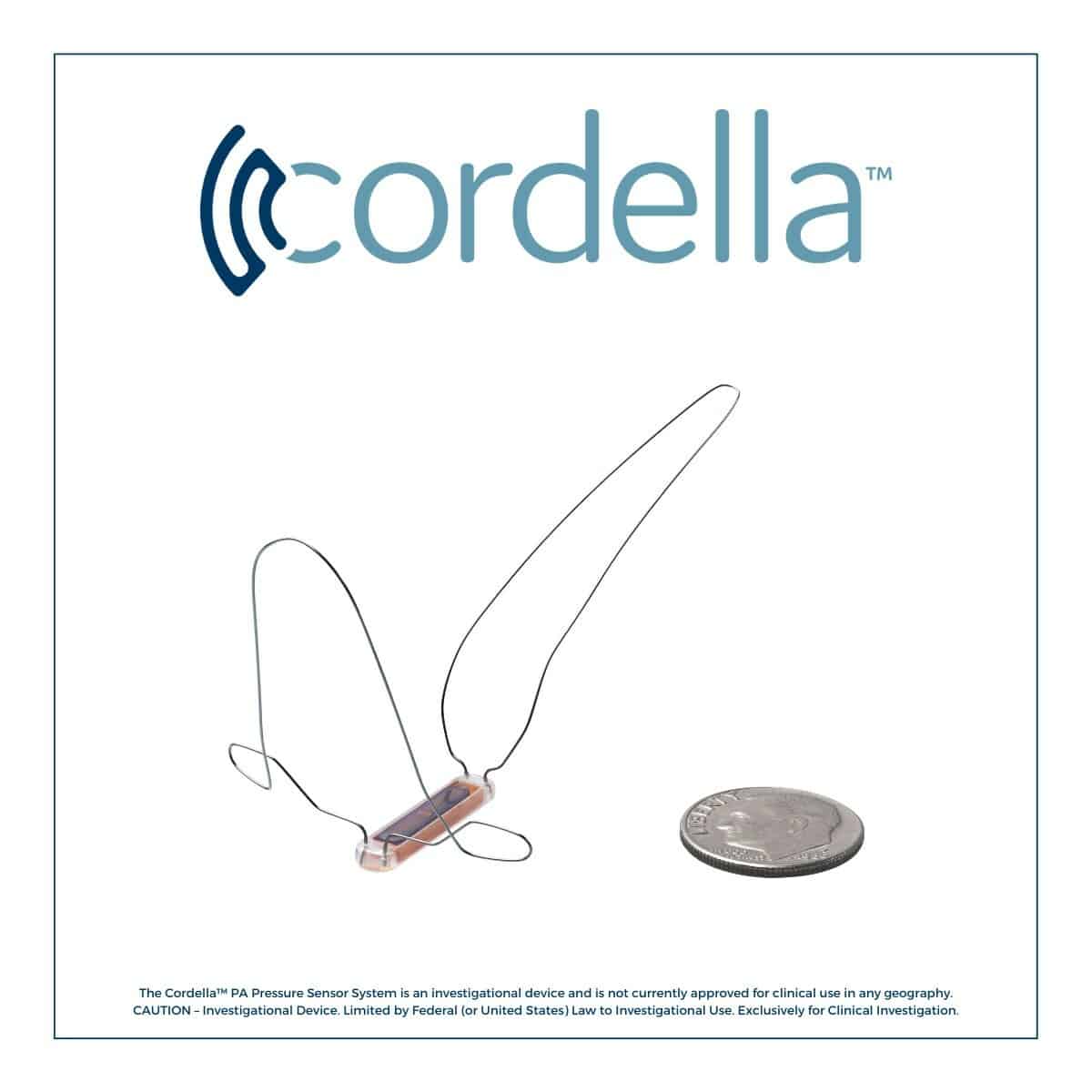
Endotronix innovates at the intersection of Medtech & Digital Health to improve care for people living with heart failure (HF). The comprehensive Cordella solution enables proactive, data-driven HF management that engages patients, reduces and prevents congestion, and improves outcomes. The Cordella PA Sensor is an implantable pulmonary artery (PA) pressure sensor which directly measures the leading indicator of congestion, allowing early, targeted therapy. The Cordella HF System is a patient health management platform, which combines comprehensive vital sign data from non-invasive devices to support patient-clinician engagement and care decisions. Combining trended insights, the versatile and scalable Cordella enhances current clinical practice and supports guideline-based care across the entire HF continuum. Learn more at www.endotronix.com.
The Cordella™ PA Pressure Sensor System is an investigational device and is not currently approved for clinical use in any geography.
CAUTION – Investigational Device. Limited by Federal (or United States) Law to Investigational Use. Exclusively for Clinical Investigation.
The Cordella HF System, without the sensor, is available for commercial use in the U.S. and E.U.
Cautionary Statement Regarding Forward-Looking Statements
This press release may contain predictions, estimates, or other information that might be considered forward-looking statements. Such forward-looking statements are not a guarantee of future performance.